COVID Study
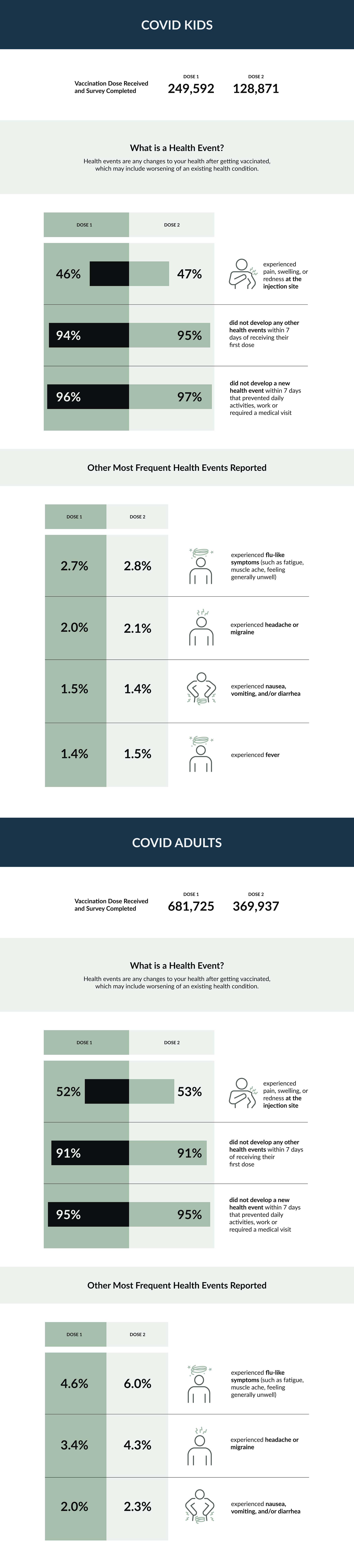
The purpose of this study is to find out how often health events occur after the primary series of the COVID-19 vaccine
Location: British Columbia, Alberta, Ontario, Quebec, Nova Scotia, PEI, Yukon and the Northwest Territories

Study participants from COVID-19 vaccine naïve populations (e.g. approval of COVID-19 vaccination for a new age group) were asked to complete up to 4 online surveys after a primary series of COVID-19 vaccine (one week after each dose, and 6 months after the last dose).
COVID-19 vaccine naïve participants were sent an email at 1 week after each primary series dose, and 6 months following the last primary series dose inviting them to respond to a short survey. Participants were asked questions regarding the occurrence of a list of signs and/or symptoms following immunization.
For CANVAS-COVID publications please follow these links:
